-
Adopt
-
Veterinary Care
Services
Client Information
- What to Expect – Angell Boston
- Client Rights and Responsibilities
- Payments / Financial Assistance
- Pharmacy
- Client Policies
- Our Doctors
- Grief Support / Counseling
- Directions and Parking
- Helpful “How-to” Pet Care
Online Payments
Emergency: Boston
Emergency: Waltham
Poison Control Hotline
-
Programs & Resources
- Careers
-
Donate Now
 By Jessica Riehl, DVM, DAVDC
By Jessica Riehl, DVM, DAVDC
Dentistry and Oral Surgery/Medicine
angell.org/dentistry
dentistry@angell.org
617-522-7282
Periodontal disease affects most of our veterinary patients. In the majority of situations, periodontal bone loss will be in a horizontal fashion. Unfortunately, opportunities to amend horizontal bone loss are not possible which is what leads to the necessity of tooth extraction. There are select situations where bone support for the tooth root can be restored, such as areas where the bone defect is vertical (infrabony) in nature.
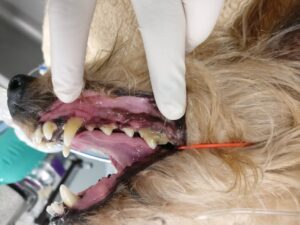
Figure 1: 9.5 year-old MN Dachshund presenting for routine dental procedure. Note the moderate calculus accumulation and seemingly normal appearance to the maxillary canine tooth from the buccal perspective.
Guided tissue regeneration (GTR) is a form of periodontal surgery which is performed to reconstruct the periodontium, in efforts to restore the anatomy and health status of a tooth. The aim is to have selective cell repopulation of specific tissues of the periodontium which have been lost to disease. The four normal components of the periodontium consist of the alveolar bone, gingiva, cementum and periodontal ligament cells. The goal is to repopulate the area with periodontal ligament cells which can redevelop cementum on the root surface and restore healthy attachment. Indications for this treatment include when the vertical bone defect is two- or three-walled in nature or class 2 furcations. For success of the procedure, not only must the surgery be executed, but maintenance of oral health will be crucial. Owners should be informed of the importance of their participation in outcome of the procedure and the follow-up which is involved. With their commitment, proper patient selection can be performed.
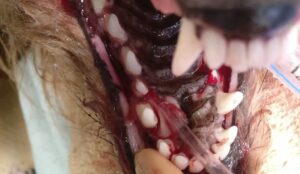
Figure 2: Same patient. Periodontal probing revealed a 6 mm pocket on the palatal and mesial aspects of the left maxillary canine tooth. A flap has been created and retracted. The root surface and pocket have been subjected to open root planning. A 3 walled infrabony pocket is present.
Contraindications for guided tissue regeneration would include a severely loose tooth, compromised health status of the patient (due to anesthesic follow-ups), lack of client compliance, lack of acceptance of home care by the pet, or complete furcational involvement of a multirooted tooth.
The most common location for guided tissue regeneration to be performed is at the palatal aspect of the maxillary canine teeth. This defect is typically 3 walled in nature and the palatal pocketing can be the only problem with the tooth. Other locations where GTR may commonly be performed include mesial and distal to the mandibular first molar, and mesial to the maxillary fourth premolar.
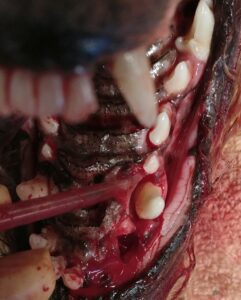
Figure 3: Same patient. Periodontal probing revealed a 7 mm pocket on the palatal aspect of the right maxillary canine tooth and a 6 mm pocket on the mesial aspect. Infrabony pocket after open root planning. The right maxillary incisors were extracted due to end stage periodontal disease.
I will use the palatal defect at the maxillary canine tooth for a description of the procedure due to the majority of typical cases falling in this description. Periodontal probing of the tooth should confirm absence of an oronasal fistula and dental radiographs should show no evidence of endodontic disease or tooth resorption.
A full thickness periodontal flap is created incising from the distal aspect of the maxillary third incisor, around the palatal aspect of the canine tooth, extending to the mesial aspect of the first premolar. The mucogingival flap is elevated from the palatal aspect of the canine tooth and retracted for better visualization. The tooth root and bone defect are prepared by performing root planing and subgingival curettage to removal all calculus and plaque, smooth the cementum, and debride any epithelial, connective or granulation tissue that is present. The area is flushed with saline or water and chlorhexidine flushing of the surgical site directly should be avoided due to its potential to adversely affect the growth of periodontal cells. A graft material is placed in the bony defect to the margin of the surrounding bone, leaving enough room between the graft material and the cervical region of the tooth to restore biologic width (2mm). Over the surface of this graft a custom-fit membrane is placed. The membrane acts to serve as a physical barrier between the root surface and the gingival flap in order to deter epithelium or connective tissue from abutting the root surface. Ideally we are guiding progenitors of periodontal ligament cells to be the primary cells to restore the periodontium. If alveolar bone cells are first to take residence, this can lead to ankylosis. If gingival epithelial cells repopulate the root surface, the periodontal pocket will recur and there will be re-establishment of the diseased state.
Bone grafting materials can be either osteoinductive or osteoconductive. The optimal material would be an autogenous bone graft which could be harvested from the same patient and would thus be osteogenic in nature as it contains osteoprogenitor cells to induce new bone formation. The challenge of obtaining an autogenous graft (increased technical skill, comorbidity of secondary surgical site, increased anesthetic time) often leads us to use other materials such as allografts or alloplasts. Allografts are harvested from one individual and donated to another individual of the same species. There are commercial products available which have been demineralized and freeze-dried to reduce or eliminate antigenicity. These allografts are both osteoinductive and osteoconductive indicating that they are able to stimulate mesenchymal cells to differentiate and begin bone formation and can provide scaffolding for the growth of new bone respectively. A third option for osteoconduction only, would be alloplasts such as synthetic particulate grafts. These materials do not have any cellular or biologic qualities but act only as a scaffold to form new bone.
Different options exist for the membrane including resorbable or non-resorbable materials. Commonly in veterinary medicine, a “membrane” of a doxycycline impregnated absorbable polymeric carrier is used as the barrier membrane. Another option for custom-fit membrane is a demineralized freeze-dried cortical bone flexible allograft.
Following a 2 week healing period, a conscious recheck should be performed and the owners may then begin to start oral home care. Daily tooth brushing is recommended in addition to other VOHC accepted products (chew treats, water additive, plaque prevention barrier). An anesthetized oral exam should be completed in 4-6 months to permit periodontal probing and dental radiograph to evaluate success of the GTR procedure demonstrated by decreased periodontal pocketing and reduction of bony defect. A full supra- and sub gingival scaling in addition to polishing should be performed to remove the plaque biofilm and any calculus. After this point in time, owners must stay committed to oral home care and follow-up oral assessments should be performed on an annual basis. With proper execution, guided tissue regeneration and bone augmentation can lead to the retention of teeth and improved oral health for our veterinary patients.
Additional reading:
- Rice CA, Snyder CJ, Soukup JW. Use of an autogenous cortical graft in combination with guided tissue regeneration for treatment of an infrabony defect. J Vet Dent 2012, 29(3)
- Stepaniuk KS, Gingerich W. Evaluation of an osseous allograft membrane for guided tissue regeneration in the dog. J Vet Dent 2015, 32(4)
- Gingerich W, Stepaniuk KS. Guided tissue regeneration for infrabony pocket treatment in dogs. J Vet Dent 2012, 29(1)