-
Adopt
-
Veterinary Care
Services
Client Information
- What to Expect – Angell Boston
- Client Rights and Responsibilities
- Payments / Financial Assistance
- Pharmacy
- Client Policies
- Our Doctors
- Grief Support / Counseling
- Directions and Parking
- Helpful “How-to” Pet Care
Online Payments
Emergency: Boston
Emergency: Waltham
Poison Control Hotline
-
Programs & Resources
- Careers
-
Donate Now
 By Courtney Peck, DVM, DACVECC
By Courtney Peck, DVM, DACVECC
angell.org/emergency
MSPCA-Angell West
781-902-8400
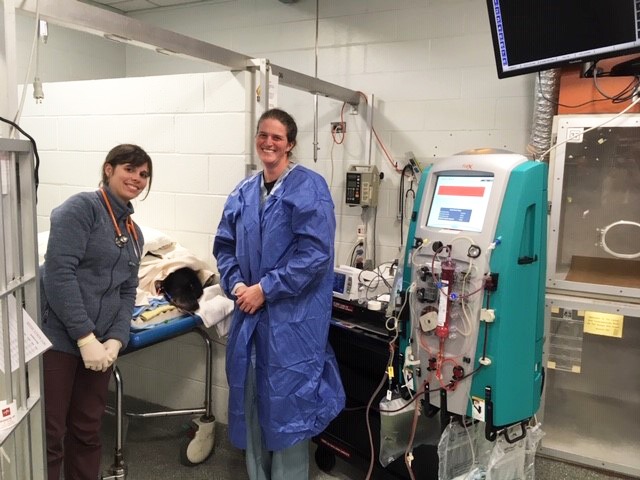
Historically, in both human and veterinary medicine, the use of hemodialysis and other extracorporeal therapies has focused mainly on treating patients with renal failure and immune-mediated diseases who have not responded to traditional medical management. Use of extracorporeal therapies for specific toxicities is still fairly early in its development and acceptance as an available treatment modality; however, an increasing number of veterinary facilities have developed extracorporeal therapy programs, creating increased access to and continued knowledge and applicability of these therapies. Recent advances in the use of extracorporeal therapies have expanded our ability to successfully manage toxicities that previously may have been fatal. As with many advances in veterinary medicine, we gain much of our theoretical knowledge from our counterparts in human medicine. Extracorporeal therapies have gained wide acceptance in human medicine in treating toxicities and drug/medication overdoses, and this application is now growing in veterinary medicine as well.
Currently, extracorporeal therapy includes four main modalities: intermittent hemodialysis (IHD), chronic renal replacement therapy (CRRT), plasmapheresis/total plasma exchange (TPE), and hemoperfusion (HP). Each of these modalities uses different principles to produce an effect. IHD is the most commonly used modality in human medicine for toxin removal; however, dialyzer filters are becoming progressively more effective, enabling other modalities to also be utilized. Specific properties of individual toxins (molecular weight; degree of protein binding; methods of endogenous clearance; and pattern of toxin distribution throughout the body [volume of distribution or VD]) determine which modality would be most effective.
 Hemodialysis relies on the process of diffusion, where molecules move from a higher concentration to a lower concentration across a semi-permeable membrane. Hemodialysis provides effective removal of small molecular weight toxins (< 500 Da). Ideal characteristics of toxins removable by hemodialysis are a low volume of distribution (VD) and a low percentage of protein binding. Over the last few decades, improvements in dialysis membranes have advanced to allow for transport of larger molecules as well as highly protein-bound molecules, increasing the utility of this modality in effective toxin therapy. Case reports in veterinary literature have described the use of IHD in successfully treating baclofen (Torre 2008), ethanol (Keno 2011), and lily (Berg 2007) intoxications.
Hemodialysis relies on the process of diffusion, where molecules move from a higher concentration to a lower concentration across a semi-permeable membrane. Hemodialysis provides effective removal of small molecular weight toxins (< 500 Da). Ideal characteristics of toxins removable by hemodialysis are a low volume of distribution (VD) and a low percentage of protein binding. Over the last few decades, improvements in dialysis membranes have advanced to allow for transport of larger molecules as well as highly protein-bound molecules, increasing the utility of this modality in effective toxin therapy. Case reports in veterinary literature have described the use of IHD in successfully treating baclofen (Torre 2008), ethanol (Keno 2011), and lily (Berg 2007) intoxications.
CRRT uses convection, which involves the movement of solvent and solutes across a semi-permeable membrane according to a pressure gradient (also known as solvent drag). The efficacy of convection is mainly dependent on the size of the dialyzer membrane pores. It has similar abilities to IHD in terms of VD and percentage of protein binding of a toxin. The main advantage of continuous techniques is their ability to remove fluid and solute over a prolonged period of time. Both blood flow and dialysate flow rates are lower in continuous therapies, resulting in a lower clearance of a toxin over a similar time period compared with IHD. These modalities are commonly used in hemodynamically or otherwise unstable patients. Clearance of middle and large solutes is greater with continuous therapies than with IHD. Slower therapies also can prevent a “rebound” effect, where plasma toxin levels will increase after a treatment as the toxin moves from the extravascular space into the intravascular compartment.
Therapeutic plasma exchange (TPE) or plasmapheresis is another extracorporeal modality that can be used for some toxicities. In this process, plasma is separated from the other blood components (either via centrifugation or across a membrane filter) and removed from the patient. It is replaced with a combination of donor plasma, colloids and crystalloid fluid to maintain hemodynamic stability. Because TPE can remove essentially all substances from plasma including proteins, it is effective in removing toxins which are large molecules as well as those that are highly protein bound. Toxins tend to be small to middle-sized molecules, so this modality is less frequently used in treating toxicities. This modality has been used with success in Amanita phalloides, vincristine, and cisplatin toxicities in humans. In veterinary medicine, TPE has been reported in the management of meloxicam (Walton 2017), carprofen (Kjaergaard 2018), and ibuprofen (Walton 2017) toxicities.
 Hemoperfusion (HP) uses specialized filters (most commonly charcoal or other specific resins/ polymers) that adsorb toxic molecules directly from the blood as it passes over them. HP will decrease the concentration of specific toxins in the blood, reducing the severity and/or duration of toxicity. This modality is effective in removing toxins with a low VD and higher degree of protein binding as well as lipophilic toxins (such as permethrins/pyrethrins, ivermectin). Molecular size is less of a consideration in this modality, unless the toxin exceeds 5000 Da, which markedly reduces clearance. These specialized filters can be incorporated into circuits and machines used by other extracorporeal therapies (IHD, CRRT). In human medicine, interest in this modality has decreased over time, in favor of hemodialysis, due to various factors (rapid saturation of the filter requiring multiple filters per treatment, binding of non-toxic substances such as blood cells, electrolytes and glucose). In veterinary medicine, HP has recently gained increased interest, and there are a few case reports describing its use (Segev, 2018 and Tauk, 2016).
Hemoperfusion (HP) uses specialized filters (most commonly charcoal or other specific resins/ polymers) that adsorb toxic molecules directly from the blood as it passes over them. HP will decrease the concentration of specific toxins in the blood, reducing the severity and/or duration of toxicity. This modality is effective in removing toxins with a low VD and higher degree of protein binding as well as lipophilic toxins (such as permethrins/pyrethrins, ivermectin). Molecular size is less of a consideration in this modality, unless the toxin exceeds 5000 Da, which markedly reduces clearance. These specialized filters can be incorporated into circuits and machines used by other extracorporeal therapies (IHD, CRRT). In human medicine, interest in this modality has decreased over time, in favor of hemodialysis, due to various factors (rapid saturation of the filter requiring multiple filters per treatment, binding of non-toxic substances such as blood cells, electrolytes and glucose). In veterinary medicine, HP has recently gained increased interest, and there are a few case reports describing its use (Segev, 2018 and Tauk, 2016).
The use of extracorporeal therapies in toxin exposure is aimed at promoting removal of exogenous or endogenous substances and/or supporting or temporarily replacing organ function. The pertinence of extracorporeal therapy in treating a toxicity should be weighed against the availability and effect of other alternate therapies, including traditional medical management. The timing of initiation of extracorporeal therapy in toxicity is critical however, as the goal is to remove the toxin and/or its metabolites as rapidly as possible, to prevent organ or cellular injury.
As with any exposure to a toxin, the patient should be stabilized, and if possible, decontamination should be attempted, if safe, immediately. Use of any specific antidotes is also recommended, if available. Once this has occurred, it is reasonable to consider the toxin and its properties, which may make it amenable to extracorporeal therapy. Additional considerations when determining whether extracorporeal therapy may be appropriate include the severity of the toxicity in terms of body systems affected (cardiovascular, respiratory, CNS, renal effects) and potential outcomes (lethal, irreversible tissue damage). If the potential risks of toxin exposure outweigh the benefits of traditional medical management, use of extracorporeal therapy should be considered. The use of these therapies may also have the potential to reduce hospitalization stay and associated costs, if a more rapid recovery is produced with extracorporeal therapy.
While supportive care remains the mainstay of management in toxicities in both human and veterinary medicine, advances in our knowledge and technical capabilities of extracorporeal therapies have allowed these modalities to increasingly become helpful in treatment in toxic exposures. Having knowledge of a toxin’s pharmacokinetic properties (molecular weight, degree of protein binding, VD), as well as an understanding of how various extracorporeal therapies work including their advantages and limitations, will allow identification of appropriate candidates for this type of therapy, and ultimately may provide favorable outcomes for patients suffering adverse effects of toxin exposure.
At Angell Animal Medical Center, we are now pleased to be able to offer extracorporeal therapies to our veterinary patients. For more information, please feel free to contact either Shawn Kearns (skearns@angell.org) or Courtney Peck (cpeck@angell.org).
Selected References:
- Monaghan KN, Acierno MJ. Extracorporeal removal of drugs and toxins. Vet Clin Small Anim 2011; 41: 227-238.
- Berg RI, Francey T, Segev G. Resolution of acute kidney injury in a cat after lily (Lilium lancifolium) intoxication. JVIM 2007; 21: 857-859.
- Keno LA, Langston CE. Treatment of accidental ethanol intoxication with hemodialysis in a dog. JVECC 2011; 21(4): 363-368.
- Torre DM, Labato MA, Rossi T, et al. Treatment of a dog with severe baclofen intoxication using hemodialysis and mechanical ventilation. JVECC 2008; 18(3): 312-318.
- Walton S, Ryan KA, Davis JL, et al. Treatment of meloxicam overdose in a dog via therapeutic plasma exchange. JVECC 2017; 27(4): 444-450.
- Kjaergaard AB, Davis JL, Acierno MJ. Treatment of carprofen overdose with therapeutic plasma exchange in a dog. JVECC 2018; 28(4): 356-360.
- Walton S, Ryan KA, Davis JL, et al. Treatment of ibuprofen intoxication in a dog via therapeutic plasma exchange. JVECC 2017; 27(4): 451-457.
- Segev G, Cowgill LD. Treatment of acute kidney injury associated with cyslosporine overdose in a dog using hemodialysis and charcoal hemoperfusion. JVECC 2018; 28(2): 163-167.
- Tauk BS, Foster JD. Treatment of ibuprofen toxicity with serial charcoal hemoperfusion and hemodialysis in a dog. JVECC 2016; 26(6): 787-792.