-
Adopt
-
Veterinary Care
Services
Client Information
- What to Expect – Angell Boston
- Client Rights and Responsibilities
- Payments / Financial Assistance
- Pharmacy
- Client Policies
- Our Doctors
- Grief Support / Counseling
- Directions and Parking
- Helpful “How-to” Pet Care
Online Payments
Emergency: Boston
Emergency: Waltham
Poison Control Hotline
-
Programs & Resources
- Careers
-
Donate Now
 By Katherine Hogan, DVM, DACVIM (Cardiology)
By Katherine Hogan, DVM, DACVIM (Cardiology)![]()
angell.org/cardiology
617-541-5038
INTRODUCTION
Patent ductus arteriosus (PDA) is the most common congenital heart defect in dogs. Retrospective studies suggest that PDAs account for ~ 11-32% of congenital heart disease in dogs; PDAs are also fairly common in cats, accounting for ~ 11% of all feline congenital heart diseases. This disease can affect dogs of all sizes, but some breeds are more predisposed, especially Maltese, toy poodle, miniature poodle, and German shepherd dogs. A genetic predisposition has been proven in the poodle and is suspected in other breeds. There is a female predisposition, as well.
The ductus arteriosus originates from the 6th aortic arch and connects the pulmonary artery to the aorta. During fetal development the pulmonary vasculature is constricted, and the ductus allows oxygenated blood to shunt from the pulmonary artery → aorta → body. At birth, lung expansion occurs and oxygen tension in the systemic vasculature increases. This leads to pulmonary vessel dilation and significantly decreased pulmonary vascular resistance. Within minutes to hours of birth, there should be contraction and physiologic closure of the ductus. A PDA results due to failure of closure of the ductus arteriosus after birth.
As a result of having a PDA, blood shunts continuously from “left to right” (aorta to pulmonary artery) due to higher aortic blood flow compared to pulmonary blood flow, resulting in continuous flow and a continuous murmur. The amount of left to right shunting depends on the size of the PDA and amount of blood allowed across the defect. The continuous left to right shunting causes the following cycle of blood flow through the heart and lungs:
Aorta ⇒ PDA ⇒ Main pulmonary artery ⇒ Lungs ⇒ Left atrium ⇒ Left ventricle ⇒ Aorta ⇒ PDA ⇒ etc.
Ultimately, if the PDA remains patent, this will lead to increased pulmonary flow → increased venous return to the left atrium (LA) and left ventricle (LV) → left-sided volume overload → LA dilation, LV dilation → left-sided congestive heart failure.
Unfortunately, if the left to right shunting PDA is not closed, this can lead to congestive heart failure by the age of 2 years old in most dogs. Additionally, there is the possibility of the PDA flow reversing; right to left shunting can occur due to increased pulmonary vasculature resistance, increased pulmonary bloodflow, and pulmonary hypertension. Treatment of right to left shunting PDAs is very different compared to left to right shunting PDA and will not be addressed during this article.
CLINICAL ASSESSMENT
Diagnosis of a PDA can be strongly suspected based on signalment, history, and physical exam findings. Patients with PDAs are often asymptomatic; patients with severe volume overload may show signs related to CHF (dyspnea/tachypnea, coughing, exercise intolerance, syncope). The murmur of a PDA is classically heard as a continuous “washing machine” murmur at the dorsal heart base. It is often heard cranially, and we recommend ausculting further up into the armpit in all dogs, especially puppies!
Definitive diagnosis of a PDA is made via echocardiography, though thoracic radiographs are also helpful in the initial workup. Thoracic radiographs may reveal left heart enlargement, pulmonary overcirculation, and often a “triple bump” appearance (due to pulmonary artery and aorta dilation plus a ductal aneurysm); finally, there may be evidence of congestive heart failure. Echocardiography of PDA will allow determination of heart enlargement and pumping function and visualization of the PDA morphology and size.

Amplatz Canine Duct Occluder (ACDO): A self-expanding nitinol mesh device, composed of a short waist that separates a flat distal disc from a cupped proximal disc. The device is designed specifically to conform to the shape of the canine ductus.
TREATMENT OPTIONS
Left to right shunting PDAs are one of the few cardiac diseases in dogs and cats in which treatment can essentially be considered curative. Surgical treatment requires a lateral thoracotomy to directly visualize, isolate, and ligate the PDA. This is commonly performed in very small dogs (e.g. dogs < 3 kg), cats, or in some larger breed dogs (especially German shepherd dogs!) with specific anatomical features of their PDA that are only amenable to surgical ligation. A less invasive option commonly performed at Angell is PDA occlusion using an Amplatz Canine Duct Occluder (ACDO). This minimally invasive procedure involves placing a catheter within the femoral artery, performing an angiogram (contrast study) of the PDA, then placing an ACDO within the PDA. This has a high success rate and overall low complication rate. Many dogs’ hearts can return to normal size, sometimes as soon as the next day!
OTHER CAUSES OF CONTINUOUS MURMURS
Though far less common, there are a few other conditions to be aware of that can also cause a continuous heart murmur. These conditions are typically a bit more challenging to diagnose and treat. Some examples include aorticopulmonary window (a “window” between the major vessels, as opposed to a ductus or vessel, which can be due to a congenital abnormality or trauma); truncus arteriosus (in which the aortic and pulmonic trunks fail to divide – quite rare), ruptured sinus of Valsalva (a.k.a. ruptured aortic sinus), and fistulae (e.g. systemic or pulmonary arteriovenous fistulae/shunts or coronary artery fistula). Either way, if a continuous murmur is ausculted, we recommend referral to a local cardiologist for further evaluation!
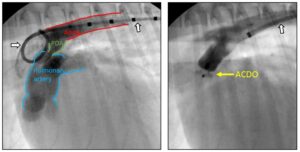
“Before and after” angiograms of a patient with successful PDA occlusion via ACDO. The image on the left shows blood flow from aorta → PDA → pulmonary artery. The image on the right reveals successful occlusion of the PDA (lack of flow from the aorta into the pulmonary artery)! (White arrow with black outline identify the catheters in each image; the catheter in the “before”/left image is a marker pigtail catheter. The catheter in the “after”/right image is a Check-Flo catheter.)
REFERENCES
- Buchanan, J.W., Patterson, D.F. Etiology of Patent Ductus Arteriosus. JVIM 2003; 17: 167-171
- Bonagura JD, Lehmuhl LB. Congenital heart disease. In Fox, Sisson Moise eds. Textbook of Canine and Feline Cardiology. 2nd WB Saunders, Philadelphia, PA 1999, p 471.
- Meurs K. Genetics of cardiac disease in the small animal patient. Vet Clinics 2010; 40: 701 – 715.
- Small Animal Cardiovascular Medicine, chapters 12, 13, 14. Online www.vin.com.
- Saunders, A.B., et al. Long-term outcome in dogs with patent ductus arteriosus: 520 Cases (1994-2009). JVIM 2014; 28: 401410
- Nguyenba TP, Tobias AH. The Amplatz canine ductal occluder: a novel device for PDA occlusion. JVC 2007: 109-117.
- Fujii, Y., et al. Arteriovenous shunts resembling patent ductus arteriosus in dogs: 3 cases. JVC 2009; 11, 147-151.