-
Adopt
-
Veterinary Care
Services
Client Information
- What to Expect – Angell Boston
- Client Rights and Responsibilities
- Payments / Financial Assistance
- Pharmacy
- Client Policies
- Our Doctors
- Grief Support / Counseling
- Directions and Parking
- Helpful “How-to” Pet Care
Online Payments
Emergency: Boston
Emergency: Waltham
Poison Control Hotline
-
Programs & Resources
- Careers
-
Donate Now
 By Susan O’Bell, DVM, DACVIM (InternalMedicine)
By Susan O’Bell, DVM, DACVIM (InternalMedicine)![]()
angell.org/generalmedicine
generalmedicine@angell.org
617-522-7282
April 2022
There should be no shortcuts when managing patients with chronic health conditions, but the internists at Angell wanted to provide you with a “highlights reel” from the last couple of years. Hopefully, some of these topics pique your interest and provide a jumping-off point to explore deeper. Let me know if I can provide you with supporting materials for any particular topics, although an appendix of resources to supplement these topics is also available at the end of these proceedings.
chronic health conditions, but the internists at Angell wanted to provide you with a “highlights reel” from the last couple of years. Hopefully, some of these topics pique your interest and provide a jumping-off point to explore deeper. Let me know if I can provide you with supporting materials for any particular topics, although an appendix of resources to supplement these topics is also available at the end of these proceedings.
Special Considerations As You Prepare Your Endocrine Patient for Anesthesia
- Patients with hyperadrenocorticism: Discontinue vetoryl (trilostane) 24 to 48 hours before surgery. Patients need some cortisol to help them cope with the stress of hospitalization and surgery. Additionally, these patients often have some degree of hypertension, so our anesthesiologists aim to keep their MAP (mean arterial pressures) higher than their normal baselines. These patients may also need more assisted ventilation, especially if they have the classic pendulous abdomen and will be in dorsal recumbency for their procedures.
- Patients with hypoadrenocorticism: Check when their last dose of mineralocorticoid was given, and try to make sure they aren’t at the end of the dosing interval. If they are close, give a dose! Regarding their glucocorticoids, these patients are usually (appropriately) on the lowest physiologic dose of steroid possible, so simply doubling the dose on the day of surgery is often not enough. There’s nothing in the literature regarding what dose is appropriate, but our anesthesiologists usually look at what dose they’re on and often default to 0.15 mg/kg dex-SP at induction. A telltale sign of an Addisonian crisis under anesthesia is hypotension refractory to traditional interventions (turn down anesthesia, fluid bolus, pressors), and they need more steroids to support them if that is the case.
- Diabetes mellitus, cat or dog: Yes, please only give ½ dose of insulin in the morning on the day of their planned anesthesia! See how recovery is going in terms of appetite, etc., before making recommendations for post-op insulin. As a rule, our anesthesiologists avoid dexmedetomidine in diabetic dogs and cats because it can further interfere with insulin/glucose ratios. Regarding monitoring, they check a blood glucose (BG) at the time of IV catheter placement and have a cut-off of lower than 350 for anesthesia. Depending on the length of the procedure, recheck BG every 45 to 60 minutes. They occasionally give regular insulin if BG is getting too high, but this is rare. The flip side of this is the RARE situation of supplementing dextrose in the diabetic with low BG when checked. Sadly, literature lacks great evidence-based practices in regards to anesthetizing diabetics.
- Thyroid disease: There has been some recent chatter about prolonged anesthetic recoveries in undiagnosed hypothyroid dogs. Troubleshoot your usual list if you have a patient with a prolonged recovery (reverse what drugs you can, get them moving, address fluid needs, etc.), but in some middle-aged to older dogs without other explanations, consider thyroid testing. Ketamine is good in hypothyroid dogs, so long as there is no significant concurrent cardiovascular disease because it provides a boost to their metabolism. Like dogs, screening T4s in cats before anesthesia is a good idea. A thyroid storm/cardiovascular collapse would be the worst complication that could have been prevented in many situations. Otherwise, do your best to have thyroid kitties as well controlled in advance as possible due to the concurrent cardiovascular complications and poor/thin body condition that goes along with hyperthyroidism. Having them as well controlled in advance is ideal, and remember you don’t need to wait too long after dose adjustments before checking a T4, so even a little “microregulation” approaching an anesthetic event could work in your favor.
Libre Application and Its Utility for Diabetic Patients
 The Freestyle Libre is an invaluable tool to help monitor various diabetic patients, including hospitalized patients with diabetic ketoacidosis, newly diagnosed patients, patients with unstable readings you are trying to stabilize, or periodic monitoring of otherwise stable patients. We recommend that clients purchase a reader rather than use their cell phone for various reasons. If the client wants to track readings on their cell phone, the sensor must be first calibrated with the Libre reader (and then the readings can be uploaded to their cell phone). Unfortunately, you cannot calibrate with a cell phone and then a reader. You may stock sensors and readers at your clinic or provide clients with a prescription to acquire a 14-day reader. In most veterinary patients (60% to 80%), the reader does NOT provide a full 14 days of readings, so try to manage client expectations appropriately and decide as a hospital your protocol for what to do when readers fall off or stop working “early.” Lately, we have tried to merely suggest using a Libre as a replacement for a traditional day-long hospitalized BG curve. It’s the 14-day label that feels misleading to clients. Tools necessary for application are the Libre sensor/applicator, a reader, clippers, alcohol, a pair of hemostats, tissue glue, and ideally some covering ranging from adhesive covers to a Suitical or ThunderShirt (see Appendix 1 for supplies). Please visit Purdue’s website for an easy to follow video on application and readings: https://vet.purdue.edu/vth/small-animal/im-endocrinology.php
The Freestyle Libre is an invaluable tool to help monitor various diabetic patients, including hospitalized patients with diabetic ketoacidosis, newly diagnosed patients, patients with unstable readings you are trying to stabilize, or periodic monitoring of otherwise stable patients. We recommend that clients purchase a reader rather than use their cell phone for various reasons. If the client wants to track readings on their cell phone, the sensor must be first calibrated with the Libre reader (and then the readings can be uploaded to their cell phone). Unfortunately, you cannot calibrate with a cell phone and then a reader. You may stock sensors and readers at your clinic or provide clients with a prescription to acquire a 14-day reader. In most veterinary patients (60% to 80%), the reader does NOT provide a full 14 days of readings, so try to manage client expectations appropriately and decide as a hospital your protocol for what to do when readers fall off or stop working “early.” Lately, we have tried to merely suggest using a Libre as a replacement for a traditional day-long hospitalized BG curve. It’s the 14-day label that feels misleading to clients. Tools necessary for application are the Libre sensor/applicator, a reader, clippers, alcohol, a pair of hemostats, tissue glue, and ideally some covering ranging from adhesive covers to a Suitical or ThunderShirt (see Appendix 1 for supplies). Please visit Purdue’s website for an easy to follow video on application and readings: https://vet.purdue.edu/vth/small-animal/im-endocrinology.php
Sensors stay in place for an average of seven days in cats. Resist the temptation to use excessive tissue glue as adverse skin reactions can and do occur. “SkinTac” wipes may help sensors stick when used before application. “SkinGrip” Freestyle Libre covers or Tegaderm patches may help sensors stay in place. Although the sensor takes readings every minute, the sensor only needs to be scanned by the reader at least once every 8 hours. The reader does tend to slightly overestimate readings when in the euglycemic or hyperglycemic range and slightly underestimate readings when in the hypoglycemic range. Overall the trends are beneficial when it comes to diabetic management. I would suggest using the LibreView site to collect and interpret data from readings. The daily reports are similar to interpreting a traditional BG curve, but with an often easier appreciation for the glucose nadir, the duration of effect of insulin, especially when using long-acting insulin or once-daily insulin administration, and average glucose levels. You are still striving for a glucose nadir of 80 – 150 mg/dL and an average BG of less than 300 mg/dL over 24 hours. Although the reader gives nearly continuous readings, you should still adjust insulin doses only every five to seven days, so if you are lucky, you may be able to get one or two adjustments from one sensor application.
Signs of FHT are commonly weight loss, goiter, a thin BCS, and heart murmur. Common diagnostics to detect FHT are total T4 and free T4. Less common diagnostics include the T3 suppression test, TRH stimulation test, and thyroid scintigraphy. Treatments for FHT include antithyroid drugs, limited iodine foods, radioactive iodine, and thyroidectomy, with each treatment having its own set of advantages and disadvantages.
“Rarer” Feline Endocrinopathies
Hyperaldosteronism: We see enough cats at Angell with this condition that it doesn’t feel uncommon anymore. If you look for it, you will find it, and it has to be considered in almost every cat with hypokalemia (<3.0mEq/L). Concurrent clin-path abnormalities include mild azotemia and CK elevations. Feline patients with K+ lower than 3.0 mEq/L either have significant renal disease or hyperaldosteronism until proven otherwise (furosemide treatment could cause potassium wasting, but this should be relatively easy to rule out). If the potassium is severely low enough to cause ventral neck flexion or if the patient also has hypertension leading to retinal changes, hyperaldosteronism is also the most likely diagnosis. Cats with K+ serum levels less than 3.0 require treatment. If possible, submit a blood sample for an aldosterone level, then begin IV supplementation if ventral neck flexion or hypoventilation are present. Begin oral supplementation if the patient is stable. Single adrenal tumors are the most likely culprit, and surgical removal may be a worthwhile pursuit. Lifelong treatment is necessary otherwise, and most need more than oral supplementation of K+ (SQF, spironolactone, address hypertension). See Appendix 2 for treatment summary and a helpful article about this condition.
this condition that it doesn’t feel uncommon anymore. If you look for it, you will find it, and it has to be considered in almost every cat with hypokalemia (<3.0mEq/L). Concurrent clin-path abnormalities include mild azotemia and CK elevations. Feline patients with K+ lower than 3.0 mEq/L either have significant renal disease or hyperaldosteronism until proven otherwise (furosemide treatment could cause potassium wasting, but this should be relatively easy to rule out). If the potassium is severely low enough to cause ventral neck flexion or if the patient also has hypertension leading to retinal changes, hyperaldosteronism is also the most likely diagnosis. Cats with K+ serum levels less than 3.0 require treatment. If possible, submit a blood sample for an aldosterone level, then begin IV supplementation if ventral neck flexion or hypoventilation are present. Begin oral supplementation if the patient is stable. Single adrenal tumors are the most likely culprit, and surgical removal may be a worthwhile pursuit. Lifelong treatment is necessary otherwise, and most need more than oral supplementation of K+ (SQF, spironolactone, address hypertension). See Appendix 2 for treatment summary and a helpful article about this condition.
Acromegaly: Acromegaly is the clinical syndrome that develops due to excess growth hormone in cats (hypersomatotropism is most commonly caused by a growth hormone-producing pituitary gland a denoma). These are more often male cats, usually 10 to 11 years old or older, with a normal body condition score (BCS) despite concurrent diabetes mellitus. Often everything is bigger (larger organs on abdominal ultrasound (AUS), larger facial features and interdental space, tongue, ventricles.) Hypersomatotropism is definitely an under-recognized disease in cats. Testing should be considered in patients with difficult-to-regulate diabetes, organomegaly of unknown cause, unexplained weight gain or changes in facial/body features, and neurologic signs. Treatment options for these cats are aggressive insulin treatment for their concurrent diabetes, radiation, hypophysectomy, or pasireotide (Appendix 3; very expensive). Whenever possible, treatment should be considered as many of these cats will do well for a long time!
denoma). These are more often male cats, usually 10 to 11 years old or older, with a normal body condition score (BCS) despite concurrent diabetes mellitus. Often everything is bigger (larger organs on abdominal ultrasound (AUS), larger facial features and interdental space, tongue, ventricles.) Hypersomatotropism is definitely an under-recognized disease in cats. Testing should be considered in patients with difficult-to-regulate diabetes, organomegaly of unknown cause, unexplained weight gain or changes in facial/body features, and neurologic signs. Treatment options for these cats are aggressive insulin treatment for their concurrent diabetes, radiation, hypophysectomy, or pasireotide (Appendix 3; very expensive). Whenever possible, treatment should be considered as many of these cats will do well for a long time!
Hyperadrenocorticism: Hyperadrenocorticism in cats is rare but should be considered in insulin-resistant diabetics or cats with fragile skin syndrome. Other symptoms are similar to those in dogs with Cushing’s (PU/PD, symmetric alopecia, infections, and peripheral neuropathy). Low-dose Dexamethasone Suppression (LDDS) is the test of choice, although extreme care must be used if fragile skin syndrome is present (consider urine testing options instead – Appendix 4). A higher dose of dexamethasone (0.1 mg/kg IV) is used than in the dog LDDS. As with dogs, the vast majority of cats with hyperadrenocorticism have the pituitary-dependent form of the disease, while 15% are diagnosed with functional adrenocortical tumors.
Starting Insulin of Choice
Cats: Glargine is our starting insulin of choice in diabetic cats. Glargine combined with a low-carbohydrate diet to minimize prandial increases in blood glucose in cats may result in the highest likelihood of remission. Twice-a-day administration is recommended. The starting dose for glargine is 0.25 U/kg of ideal body weight, if blood glucose concentration is <360 mg/dL or 0.5 U/kg if blood glucose is >360 mg/dL. Lantus Solostar (glargine) 3 mL pens are less expensive options for clients that have allowed us to continue offering glargine as a starting choice despite the recent increase in cost. It is possible to draw insulin out of the top of the pen rather than inject directly with the pen (although some clients like the pens and tiny needles and feel less like they are “giving an injection,” so keep this in mind for fearful owners).
starting choice despite the recent increase in cost. It is possible to draw insulin out of the top of the pen rather than inject directly with the pen (although some clients like the pens and tiny needles and feel less like they are “giving an injection,” so keep this in mind for fearful owners).
Another option for cats (less $): Protamine zinc insulin (PZI; ProZinc, Boehringer Ingelheim) is available for use in cats in the US and UK, having been removed from the human market in the 1990s. ProZinc has a concentration of 40 U/mL, and therefore, the use of appropriate insulin syringes must be used. Initial dose recommendations are either 1–3 U/cat (0.22–0.66 units/kg) or 0.25–0.5 units/kg ideal body weight every 12 hours, depending on the severity of clinical signs and hyperglycemia. Study results include a review of 133 diabetic cats treated with PZI (120 newly diagnosed and 13 previously treated with other insulin), showing 85% obtained good control within 45 days, and some cats able to achieve remission within ~3-4 months. (See appendix 6 for a good 2018 review article on feline diabetes). Maintaining euglycemia as much as possible throughout the day and low-carb diets are some of the more beneficial ways to achieve remission in cats, so we see no reason why you could not offer PZI as the first choice. It benefits from being FDA-approved for use in diabetic cats, and many argue that U40 syringes are easier for clients to use.
Dogs: Vetsulin vs. Neutral Protamine Hagedorn (NPH). Several Angell clinicians still use NPH to start new diabetic dogs. It has worked well for them historically, and they are most familiar with its use, but it has gotten more expensive. Many start with Vetsulin instead due to its lower cost than NPH and a very user-friendly website we can refer clients (and clinicians!) to familiarize themselves with diabetes mellitus in general. Furthermore, Vetsulin, like PZI in cats, uses U40 syringes and is FDA approved for use in dogs, and is also the AAHA Taskforce’s recommendation as a starting insulin of choice in dogs. We typically use 0.25 u/kg BID as a starting dose which is comparable to Vetsulin’s label recommendation: If twice-daily treatment is initiated, each of the two doses should be 25% less than the once-daily dose of Vetsulin®. Although the Vetsulin label suggests starting dogs at 0.5 u/kg once daily, Angell clinicians typically start twice daily.
Newer Insulins/Formulations
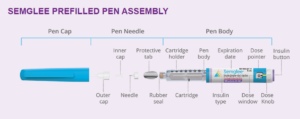 Semglee is a long-acting insulin analog that is the approved biosimilar for insulin glargine (Lantus). It is used to treat human patients with diabetes. Semglee has been proven to be as safe and effective as Lantus and provide the same health outcomes in humans. We expect it will have similar health outcomes in animals, but this has yet to be proven. Anecdotal evidence (very small numbers) suggests it works similarly in cats as our traditional Lantus option for glargine at a lower cost. Studies are in the works in cats. It is available as a pen. Since we have been using Solostar pens successfully in cats, we have had fewer issues with the rising cost of Lantus. See Appendix 8 for a helpful review of the use of pens in veterinary patients in general.
Semglee is a long-acting insulin analog that is the approved biosimilar for insulin glargine (Lantus). It is used to treat human patients with diabetes. Semglee has been proven to be as safe and effective as Lantus and provide the same health outcomes in humans. We expect it will have similar health outcomes in animals, but this has yet to be proven. Anecdotal evidence (very small numbers) suggests it works similarly in cats as our traditional Lantus option for glargine at a lower cost. Studies are in the works in cats. It is available as a pen. Since we have been using Solostar pens successfully in cats, we have had fewer issues with the rising cost of Lantus. See Appendix 8 for a helpful review of the use of pens in veterinary patients in general.
Toujeo and Tresiba: Insulin glargine 300U/ml (Toujeo®) and insulin degludec (Tresiba®) are synthetic insulin analogs. In people, Toujeo is more predictable and longer-acting compared to glargine 100U/ml (Lantus®); Tresiba is ultra-long acting with a duration of action of over 40 hours that allows a flexible daily schedule of administration. In dogs and cats, very little is known about these formulations. Tresiba has been studied in a small number of healthy cats and seems to have a much shorter duration of action in cats (about 11 hours) compared to people. Toujeo has a longer duration of action in cats (about 16 hours) and a relatively flat time-action profile. Overall, Toujeo seems the best candidate out of the currently available insulin formulation for use as a once-daily injection in both dogs and cats. However, Toujeo is not very potent, and the total daily dose might be high, making it somewhat expensive. For cats, consider 1-2 U/cat once daily to start. For dogs, start with 0.4U/kg once daily (but you might end up much higher than that due to potency issues). I would try PZI in cats before reaching for Toujeo for a number of reasons, even if only once-daily dosing is an option for clients.
insulin degludec (Tresiba®) are synthetic insulin analogs. In people, Toujeo is more predictable and longer-acting compared to glargine 100U/ml (Lantus®); Tresiba is ultra-long acting with a duration of action of over 40 hours that allows a flexible daily schedule of administration. In dogs and cats, very little is known about these formulations. Tresiba has been studied in a small number of healthy cats and seems to have a much shorter duration of action in cats (about 11 hours) compared to people. Toujeo has a longer duration of action in cats (about 16 hours) and a relatively flat time-action profile. Overall, Toujeo seems the best candidate out of the currently available insulin formulation for use as a once-daily injection in both dogs and cats. However, Toujeo is not very potent, and the total daily dose might be high, making it somewhat expensive. For cats, consider 1-2 U/cat once daily to start. For dogs, start with 0.4U/kg once daily (but you might end up much higher than that due to potency issues). I would try PZI in cats before reaching for Toujeo for a number of reasons, even if only once-daily dosing is an option for clients.
What to Feed Diabetic Cats or Dogs?
Cats: I often do not switch cat diets the same visit I start insulin. I worry that this can be too stressful for both the cat and the client. I have had success getting cats off insulin after  switching cats to low carb (≤12% metabolizable energy [ME]) and canned food diets (as supported by literature), so I do think this is a worthwhile venture. You need to use a balanced, low-carbohydrate diet–ideally one designed for diabetic cats. Canned Purina DM has one of the lowest carbohydrate contents of veterinary diets formulated for diabetes (<6% ME); highest remission rates are reported using this very low carb diet and insulin protocols aimed at achieving euglycemia throughout as much of the day as possible. You could try increasing the palatability with gravy or warming or frying if cats aren’t initially taking to it. Obesity is the most important acquired risk factor for diabetes, as overweight cats have a 4.6 times greater risk of diabetes than cats in ideal body condition. Longer-term, it is important to coach clients to achieve an ideal body weight for their diabetic cats, but most overweight cats go into remission before substantial weight loss occurs. Weight management is important for maintaining remission in many cats, though. Many low-carbohydrate diets are quite energy-dense, so you need to control the amount being fed. (Encourage clients to invest in a baby scale and give them an exact amount to feed).
switching cats to low carb (≤12% metabolizable energy [ME]) and canned food diets (as supported by literature), so I do think this is a worthwhile venture. You need to use a balanced, low-carbohydrate diet–ideally one designed for diabetic cats. Canned Purina DM has one of the lowest carbohydrate contents of veterinary diets formulated for diabetes (<6% ME); highest remission rates are reported using this very low carb diet and insulin protocols aimed at achieving euglycemia throughout as much of the day as possible. You could try increasing the palatability with gravy or warming or frying if cats aren’t initially taking to it. Obesity is the most important acquired risk factor for diabetes, as overweight cats have a 4.6 times greater risk of diabetes than cats in ideal body condition. Longer-term, it is important to coach clients to achieve an ideal body weight for their diabetic cats, but most overweight cats go into remission before substantial weight loss occurs. Weight management is important for maintaining remission in many cats, though. Many low-carbohydrate diets are quite energy-dense, so you need to control the amount being fed. (Encourage clients to invest in a baby scale and give them an exact amount to feed).
Monitor carefully as insulin requirements may decrease as cats convert to low/very low carb diets. I have also had the experience of cats being hypoglycemic on glargine without apparent clinical signs of hypoglycemia which is both reassuring and disconcerting at the same time. Try the Libre sensor for peace of mind, especially when instituting a diet change or dose adjustment.
Dogs: I am not as picky about what they eat as long as it is consistent and “good food.” If concurrent medical conditions, those nutritional needs dictate those necessitated by their diabetes. Ideally, feed two meals a day, with fewer than 10% of their calories coming through treats the rest of the day, and administer insulin 15 to 60 minutes after eating.
ACTH Stim vs. LDDS
Low-dose dexamethasone suppression tests are the test of choice to diagnose hyperadrenocorticism in dogs at Angell in conjunction with an abdominal ultrasound, although consider cost and patient compliance rather than making this a blanket rule. And honestly, it comes down to what is most practical and appealing for clients and your practice (many prefer a 1-hour test!). Choose an adrenocorticotropic hormone (ACTH) stim over LDDS in dogs who will have trouble being at the hospital for the day (i.e., lar par dog, collapsing trachea dog who gets stressed, any super stressed or challenging patient). If cost is a major factor, you may elect an ACTH stim, so you have this test performed before starting a patient on trilostane.
Trilostane Dosing
Starting doses: 1mg/kg BID vs. 2 mg/kg SID. The internists at Angell differ! Some start trilostane at about 1 to 2 mg/kg BID, but try to “round down” or go on the lower end of the dose (it all depends on the dog size and what capsule size we have). For the “BID dose preferred” group, they think that given the half-life is so short, BID just makes more sense, and certainly, BID in more serious conditions would be recommended. All agree that concurrent diabetics try to start BID, for example. The “SID starting dose” group tries 2 mg/kg once daily for six weeks before considering BID dosing.
Managing Hyperadrenocorticism
ACTH stim vs. cortisol: For monitoring, many Angell clinicians always start with an ACTH stim, and then if the patient is clinically doing well, it’s fine to do a pre-pill cortisol level for further monitoring. Essentially you are making sure this single cortisol level is not too low. If the patients are not doing well, an ACTH stim should be done. Many people still will do ACTH stim for monitoring, but these costs add up for clients, and they truthfully are probably not needed in dogs that are clinically doing well, or at least “not acting Addisonian.” Another common practice for trilostane monitoring is to run serum chemistry and cortisol two to six hours post trilostane. If this single cortisol comes back <1, then consider ACTH stim. Universally, everyone’s primary focus is on clinical signs, not cortisol values. See Appendix 5 and 6 for two opinions on interpreting “pre-pill” cortisol levels.
Clinical signs: Sadly, few treatments for Cushing’s seem really great for controlling proteinuria. Anecdotally, however, these patients don’t seem to get progressive renal disease in instances where proteinuria is caused by Cushing’s disease. If proteinuria is not “horrible” (in other words, UPC under five or six), some clinicians don’t further address the proteinuria. However, if an animal had concurrent hypertension and is still hypertensive after you treat (successfully) for Cushing’s, we would definitely recommend treatment for ongoing hypertension.
Clinical sign management is essential, but some ACTH monitoring is still required as a significant percentage of dogs will develop hypoadrenocorticism (estimated cumulative incidence of hypoadrenocorticism in a study of trilostane-treated dogs was 15% by 2 years and 26% by 4.3 years. Of the dogs that developed hypoadrenocorticism, 74% were transient in nature, and 26% were permanent.) See Appendix 7 for additional clinical sign/clin path findings when monitoring after starting trilostane.
Canine Hypothyroidism
I used to be a big believer in screening only dogs who appeared to have more classic symptoms of hypothyroidism. My justification for screening T4s has a much lower threshold than it used to. I screen all dogs with mild nonregenerative anemia, elevated cholesterol or triglycerides, or mildly elevated ALP/ALT. Whether to add just a TT4 or run a “thyroid panel” (TT4, fT4, TSH) is up to you, ease of running lab tests, and cost to the client. Most hypothyroid dogs have TT4 below your lab’s reference range, but as you know, patients with concurrent illness or exposure to certain medications (glucocorticoids, phenobarb) could have low TT4 and not have true hypothyroidism. NSAIDs do not appear to impact TT4 levels significantly. That being said, a TT4 is an excellent screening test; if the TT4 concentration is well within reference range, it is very likely the dog is euthyroid, and further thyroid testing is not required. A patient with an elevated TSH and low to low normal TT4 and fT4 should be started on levothyroxine. Up to 33% of hypothyroid dogs have normal TSH concentrations, so don’t hold out for an elevated TSH to start treatment.
Although the literature shows evidence that most dogs do well with once-daily supplementation at 0.02 mg/kg PO q24h (0.1 mg levothyroxine per 10 pounds is a general rule of thumb and I round down, especially in larger breed or obese patients), at Angell many clinicians still start twice-daily supplementation. Sometimes I decide based on the very unscientific “how real” I think the diagnosis is. You could also attempt to reduce the dosing to once-daily after clinical signs are well controlled. First, rechecks should not be before six weeks. Although steady states are reached sooner than that and TT4 could be checked as early as 10 to 14 days, clinical conditions often do not start to improve for 4 to 6 weeks, so it makes more sense to recheck both clinical conditions and labwork together at the six-plus week mark. For dogs on BID dosing, TT4s can be checked usually four to six hours after pill administration (by three hours post-pill, peak concentrations should be reached). Aim for the high end of your reference range to even a little above. Ideally, for dogs on SID dosing, trough levels are measured (just before the next pill is due). This can be tricky depending on what time of day your client administers medication.
Appendix
1. Libre section supplies.
https://www.freestyle.abbott/us-en/products/freestyle-libre-app.html
Data management site (clients can send data to you through this site): https://www.libreview.com/
FreeStyle libre coverings and skin tac wipes can be purchased here: https://skingrip.com/products/free-samples?variant=39927765598364 –
Suitical: https://www.chewy.com/suitical-recovery-suit-cats/dp/140901
2. Hyperaldosteronism treatment: https://www.mspca.org/angell_services/feline-hyperaldosteronism/
3. Pasireotide is a novel multi-receptor ligand somatostatin analog that has been shown to improve biochemical control of human patients with hypersomatotropism. Somatostatins act on the pituitary gland to inhibit growth hormone release. In addition, they can act peripherally by interfering with GH receptor binding on hepatocytes and may induce apoptosis in pituitary adenomas resulting in shrinkage. The drug is given SQ every 12-24 hours or can be administered monthly with a long-acting formulation. The main adverse effects reported are gastrointestinal upset, including soft, voluminous stools and gas-distended intestines. Cost remains a limiting factor in the use of this drug.
4. The urine cortisol:creatinine ratio (UCCR) is a useful screening test for HAC in cats as in dogs and should ideally be collected at home on two consecutive days. An alternative ‘at home’ oral dexamethasone suppression test has been suggested in dogs and may be considered in cats, especially with skin fragility syndrome. The owner is instructed to collect two urine samples from the patient on two consecutive mornings and store them in the refrigerator. After collecting the second urine sample, the owner should administer three doses of dexamethasone (0.1 mg/kg/dose) orally at eight-hour intervals. Urine is collected on the morning of the third day, and UCCR is determined on all three samples. The first two urine samples establish the diagnosis of hyperadrenocorticism; both results must be abnormal. If both values are abnormal, then the average of the two values is used as the ‘baseline’ value and compared with the third value obtained after dexamethasone administration. A pituitary-dependent hyperadrenocorticism (PDH) diagnosis is established if the UCCR result from the third urine sample is less than 50% of the ‘baseline’ value. Patients failing to meet these criteria could have either adrenal-dependent hyperadrenocorticism (ADH) or PDH. – Courtesy of Shawn Kearns, DVM, DACVIM
Analyzing Pre-pill Cortisol Levels
In clinically well dogs in whose clinical signs are well controlled and prepill cortisol levels are as follows:
- <2 μg/dL: An ACTH stimulation test should be performed (or the dose decreased).
- >2 μg/dL: It is likely safe to continue the current dose.
In clinically well dogs with persistent clinical signs of HAC and prepill cortisol levels are as follows:
- <2 μg/dL: History and clinical signs should be re-evaluated, and an ACTH stimulation test should be performed.
- >2 and <5 μg/dL: An ACTH stimulation test can be considered before increasing the dose, or doses being given once daily can be split and given twice daily; increasing the dose can be considered if cortisol levels are >3 μg/dL.
- >5 μg/dL: It is likely safe to increase or split the dose
6. Summary article for feline diabetes: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6053045/
7. Macroadenoma syndrome develops in dogs with pituitary-dependent HAC when the pituitary tumor begins to grow and applies pressure to surrounding structures in the brain. Neurologic signs associated with macroadenoma syndrome occur in approximately 10% to 30% of dogs with pituitary-dependent HAC, with most cases showing signs after initiation of treatment. The most common signs are behavior changes (e.g., dullness, restlessness, loss of interest in normal activities, disorientation, pacing) and decreased appetite. Initial or mild signs may be dismissed as changes associated with normal aging or as side effects of trilostane. You should keep the progression of pituitary macroadenoma in the back of your mind. Advanced imaging is necessary for antemortem diagnosis of pituitary macroadenoma, and treatment most commonly involves radiation therapy (they can do quite well if clients can afford it and the patient is a good candidate for it). Electrolytes: Almost quite predictably, potassium will be elevated +/- slightly low sodium. We see hyperkalemia with enough frequency that it would seem almost odd for it not to be at least slightly elevated. A mild increase is “ok” if the patient is feeling well. Still, with any concurrent signs of illness or concurrent potassium-sparing medications such as potassium-sparing diuretics, ACE inhibitors, and angiotensin-receptor blockers [e.g., telmisartan], you could cause more serious issues.
8. Helpful/thorough review of the use of insulin pens in veterinary patients: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6067590/