-
Adopt
-
Veterinary Care
Services
Client Information
- What to Expect – Angell Boston
- Client Rights and Responsibilities
- Payments / Financial Assistance
- Pharmacy
- Client Policies
- Our Doctors
- Grief Support / Counseling
- Directions and Parking
- Helpful “How-to” Pet Care
Online Payments
Emergency: Boston
Emergency: Waltham
Poison Control Hotline
-
Programs & Resources
- Careers
-
Donate Now
 Pamela Mouser, DVM, MS, DACVP
Pamela Mouser, DVM, MS, DACVP
Anatomic Pathologist
angell.org/lab
617-541-5014
For many conditions amenable to biopsy, histopathology affords a relatively straight-forward diagnosis, and treatment can ensue when the biopsy report is in hand. However, on some occasions the case necessitates an additional step in the diagnostic pathway to more accurately characterize the process: immunohistochemistry. Fortunately, immunohistochemistry is typically performed on the original formalin-fixed biopsy tissue, and does not require additional samples to be collected from the animal. This article will briefly review immunohistochemistry (IHC) technique and some common uses in veterinary pathology.
Immunohistochemistry Basic Technique
Two detailed reviews and a book chapter nicely describe IHC technique in veterinary pathology, and the following is a relatively superficial summary of the process, using these three references.1-3
As the name implies, immunohistochemistry makes use of antibodies (immuno-) applied to biopsy sections on a glass slide (-histo-) followed by a chemical reaction to make the antibodies visible under the microscope (-chemistry). Antibodies for IHC are generated by immunizing an animal, typically a mouse or rabbit, with “pure” antigen to generate an immune response. The polyclonal antibodies harvested in this way may react to different regions, or epitopes, of the target antigen, but may also include irrelevant antibodies. While the broad reactivity of polyclonal antibodies may increase the likelihood of targeted antibody-antigen binding, it may increase the risk of background or false-positive labeling due to cross-reactivity with non-target antigens. A more labor-intensive technique using mouse myeloma cells is employed to develop monoclonal antibodies, which bind a single epitope and are therefore highly specific for the antigen in question.
Formalin fixation of biopsy samples functions to cross-link proteins in order to preserve cellular integrity and prevent autolysis. However, this cross-linking may alter the protein’s structure, and ultimately obscure it from recognition by an antibody. An early step in the immunohistochemical procedure is antigen retrieval, which employs heat or chemicals in an attempt to reverse the morphologic changes to the proteins and therefore better expose the target epitopes.
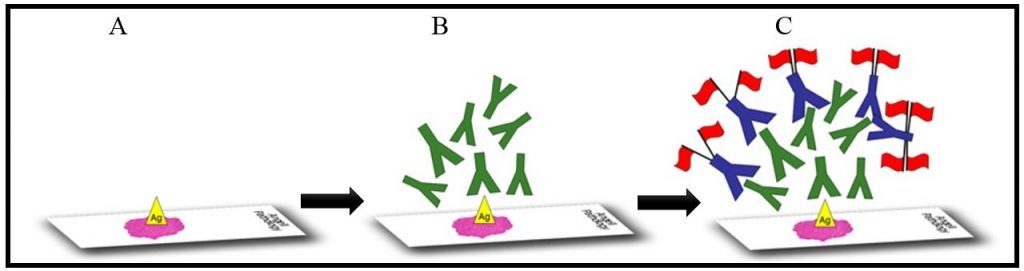
Figure 1. Step A: An unstained histology slide is routinely prepared and antigens are “unmasked” using antigen retrieval. Step B: Primary antibodies (illustrated in green), which target epitopes on the antigen of interest, are applied to the sample. Step C: A labeling system which often employs secondary antibodies (in blue) is added next, so the primary antigen-antibody reaction can be visualized under the microscope.
Similar to other immunologic tests, such as ELISA, a label is required to visualize the antigen-antibody reaction. With immunohistochemistry, this is most often accomplished using a label attached to secondary antibodies which recognize the unbound “tail” of the primary antibodies. Figure 1 depicts the major steps of the IHC process.
Both false positive and false negative results can occur with immunohistochemistry: false positives as a result of aberrant immunoreactivity (causing the appearance of positive labeling where there should be none) and false negatives due to the inability of the antibody to recognize/bind the specific epitope (may occur if target antigen remains cross-linked, specimen is poorly-fixed, or sample was treated with strong acid for decalcification as examples). To aid in the interpretation of IHC, positive and negative control samples are run in parallel with diagnostic samples. In addition, often a panel of markers is employed in a given case to provide redundancy. For example, a panel of melanocytic markers applied to amelanotic melanomas was shown to have greater sensitivity (93.9%) than the most commonly-used single marker, Melan-A, alone (81.6%).4 Laboratories offering IHC should have good quality control practices, adequate time spent on validation, and a lot of patience!
Diagnostic Utility of Immunohistochemistry
Immunohistochemistry is most routinely applied to oncologic and infectious disease cases. For neoplastic processes, IHC may be recommended for tumors which cannot be adequately classified based on clinical information, site, cytology (when available), and histomorphology. “Adequately classified” may not always mean that the exact cell of origin is determined; rather, IHC may place the neoplasm into a narrower category to better guide treatment options and help dictate prognosis.
Case Example: A 9-year-old Boston terrier was presented to Angell with an enlarged, firm right mandibular lymph node. Cytology of a fine needle aspirate showed neoplastic cells, although lineage was not definitively determined: round cell neoplasm was favored but poorly-differentiated carcinoma/sarcoma could not be excluded. Diagnostic imaging (CT) of the head and thorax confirmed moderate to severe enlargement of the mandibular lymph nodes bilaterally, as well as enlargement of the medial retropharyngeal lymph nodes bilaterally and the right superficial cervical lymph node. Importantly, a primary mass lesion was not detected with imaging, although tonsils appeared “mildly thickened” bilaterally. The right mandibular and right retropharyngeal lymph nodes were surgically removed, and histopathology confirmed the presence of a neoplastic cell population partially effacing both lymph nodes. Immunohistochemical markers employed in this case included cytokeratins, which are proteins found in epithelial cells; vimentin, an intermediate filament found in fetal and mesenchymal cells; and CD18, a leukocyte adhesion molecule. The goal of this panel was to determine if the neoplasm, presumed to be metastatic, could be classified as a carcinoma, sarcoma, or round cell neoplasm. Figure 2 illustrates the pattern of immunolabeling in atypical cells, which indicated an epithelial origin for the neoplasm (metastatic carcinoma).
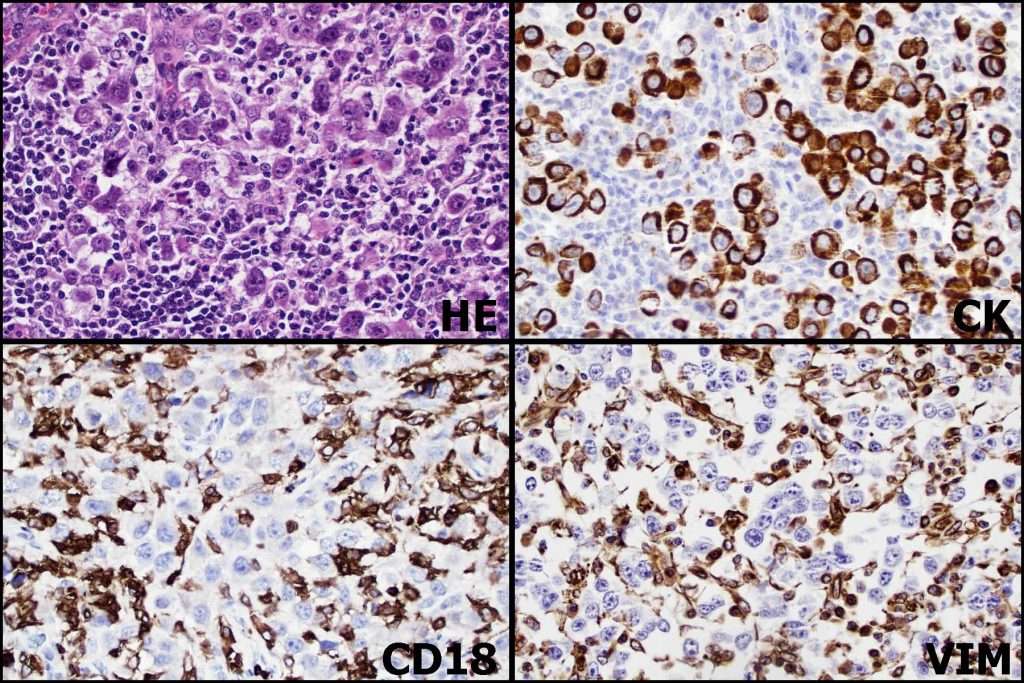
Figure 2: High magnification photomicrographs of the right retropharyngeal lymph node from a Boston terrier. The routinely-stained section (HE) shows large atypical cells effacing lymph node parenchyma. They are a paler purple color when compared to the dark blue clustered small lymphocytes to the lower left of the image. Dark brown positive cytokeratin (CK) immunolabeling is strong in large atypical cells but negative in lymphoid and stromal cells (pale blue). Conversely, CD18 and vimentin show strong brown immunolabeling of leukocytes or leukocytes and stromal cells, respectively, in the lymph node. Atypical cells are negative (light blue). IHC performed at Purdue University Animal Disease Diagnostic Laboratory.
For many neoplastic processes in which IHC is useful, cell morphology allows the tumor to be categorized as epithelial, mesenchymal, or round cell origin, but additional classification is desired. For example, treatment may differ for a sarcoma arising in the gastrointestinal tract whether the lesion arises from smooth muscle (leiomyosarcoma) or interstitial cells of Cajal (gastrointestinal stromal tumor or GIST). These lesions may be indistinguishable from one another based on histopathology alone, but can be separated using IHC markers targeting the specific cell types.5,6 In cases of lymphoma, IHC can be applied to immunophenotype the neoplasm as T- or B-cell, which has prognostic significance. Finally, some IHC markers are applied to known tumor types, such as mast cell tumors, as a way to better predict the behavior of the neoplasm (proliferation/prognostication markers).7
Some organisms are not readily detected in routinely stained slides, but can be highlighted using IHC. Antibodies against a wide range of infectious agents are available, including viruses, bacteria, fungi, and protozoa. We have found immunohistochemistry to be useful in cases of canine distemper virus, feline infectious peritonitis (FIP) virus, and papillomavirus infections here at Angell. One benefit of IHC for infectious agents over other modalities is the ability to visualize the distribution of antigen within the tissue. For example, positive immunolabeling for FIP should be localized in macrophages within inflammatory foci (granulomas).8,9 Similarly, the morphology of organisms (such as crescent-shaped protozoal tachyzoites) should be evident with a positive reaction. These features are both helpful and necessary in interpreting a positive/negative reaction.
The repertoire of antibodies available for IHC in animals is ever-expanding, providing a broader range of options to characterize cell populations, assess cellular proliferation, or identify infectious agents. We should not hesitate to pursue this ancillary test but, as with any diagnostic test, should always interpret results in light of the whole clinical picture including cytology and histopathology.
Take-home points
- Immunohistochemistry is a multi-step process, typically performed on formalin-fixed biopsy tissue, which requires careful antibody selection, validation, and a bit of finesse
- Antibodies against cellular proteins are useful to identify/classify neoplastic processes, guide treatment, and aid in prognostication of some tumor types
- A variety of infectious agents may be visualized within histopathologic lesions using IHC
References
- Ramos-Vara JA. Technical aspects of immunohistochemistry. Vet Pathol 2005;42:405-26.
- Ramos-Vara JA, Borst LB. Immunohistochemistry: fundamentals and applications in oncology. In: Meuten DJ ed. Tumors in domestic animals 5th Ames, IA: John Wiley & Sons Inc. 2017:44-87.
- Ramos-Vara JA, Miller MA. When tissue antigens and antibodies get along: revisiting the technical aspects of immunohistochemistry—the red, brown, and blue technique. Vet Pathol 2014;51:42-87.
- Smedley RC, Lamoureau J, Sledge DG, Kiupel M. Immunohistochemical diagnosis of canine oral amelanotic melanocytic neoplasms. Vet Pathol 2011;48:32-40.
- Frost D, Lasota J, Miettinen M. Gastrointestinal stromal tumors and leiomyomas in the dog: a histopathologic, immunohistochemical, and molecular genetic study of 50 cases. Vet Pathol 2003;40:42-54.
- Hayes S, Yuzbasiyan-Gurkan V, Gregory-Bryson E, Kiupel M. Classification of canine nonangiogenic, nonlymphogenic gastrointestinal sarcomas based on microscopic, immunohistochemical, and molecular characteristics. Vet Pathol 2013;50:779-88.
- Webster JD, Yuzbasiyan-Gurkan V, Miller RA, Kaneene JB, Kiupel M. Cellular proliferation in canine cutaneous mast cell tumors: associations with c-KIT and its role in prognostication. Vet Pathol 2007;44:298-308.
- Kipar A, Bellmann S, Kremendahl J, Kohler K, Reinacher M. Cellular composition, coronavirus antigen expression and production of specific antibodies in lesions in feline infectious peritonitis. Vet Immunol Immunopathol 1998;65:243-57.
- Tammer R, Evensen O, Lutz H, Reinacher M. Immunohistological demonstration of feline infectious peritonitis virus antigen in paraffin-embedded tissues using feline ascites or murine monoclonal antibodies. Vet Immunol Immunopathol 1995;49:177-82.