-
Adopt
-
Veterinary Care
Services
Client Information
- What to Expect – Angell Boston
- Client Rights and Responsibilities
- Payments / Financial Assistance
- Pharmacy
- Client Policies
- Our Doctors
- Grief Support / Counseling
- Directions and Parking
- Helpful “How-to” Pet Care
Online Payments
Emergency: Boston
Emergency: Waltham
Poison Control Hotline
-
Programs & Resources
- Careers
-
Donate Now
 By Daniela Vrabelova Ackley, DVM, MS, DACVIM angell.org/internalmedicine
By Daniela Vrabelova Ackley, DVM, MS, DACVIM angell.org/internalmedicine
dvrabelova@angell.org
781-902-8400
MSPCA-Angell West, Waltham
In an age of advancing technologies, the management of urolithiasis poses a unique opportunity to advance veterinary care. In 2016, a panel of veterinary internists with special interest in urology published a consensus statement in the Journal of Veterinary Internal Medicine to offer suitable contemporary strategies in the management of urolithiasis.1 The panelists recognized that the availability of minimally invasive technology or willingness to travel to centers of expertise may be a limitation, but the recommendations were selected in the patient’s best interest as if all options were available. These recommendations apply to stable patients.
Treatment of lower urinary tract (bladder and urethra) uroliths
- Struvite uroliths should be medically dissolved while treating associated UTI, as this is a highly effective approach, unless dissolution foods are not tolerated, or the uroliths cannot be adequately bathed in urine (extremely large uroliths, or due to urinary obstruction). Struvites are usually produced by urease-producing bacteria, however, sterile struvites are also amenable to dissolution (usually in 2-5 weeks). Urethral obstruction as a potential risk of dissolution has not been reported in the veterinary literature.
- Urocystoliths associated with clinical signs should be removed by minimally invasive procedures including medical dissolution, voiding urohydropropulsion, or basket retrieval, if they are small enough to pass through the urethra. Otherwise, intracorporeal laser lithotripsy or percutaneous cystolithotomy can be used. Authors recommend avoiding cystotomy to decrease the risk of suture-induced uroliths, length of hospitalization and anesthesia time, and the number residual stones due to improved visualization. Dissolution of urate uroliths should be attempted before removal and can usually be accomplished within 4 weeks by feeding a purine-restricted, alkalinizing, diuretic diet, along with allopurinol therapy (15 mg/kg PO q12h). Dissolution has not been possible in dogs and cats with uncorrected liver disease. Similarly, dissolution of cystine uroliths can be achieved by feeding an alkalinizing, protein-restricted diet with the addition of 2-mercaptopropionylglycine (Thiola) at 15-20mg/kg PO q12h.
- Nonclinical nondissolvable urocystoliths unlikely to cause urinary obstruction (due to their size) do not require removal but monitoring and appropriate client education about the signs of urinary obstruction.
- Nonclinical urocystoliths likely to cause obstruction (diameter similar to the urethra) should be removed by minimally invasive procedures.
- Urethroliths should be managed by intracorporeal lithotripsy and basket retrieval, which is a safe, quick, and highly effective procedure.
- Urethral surgery is discouraged and considered a salvage procedure. Urethroliths should be repositioned by retrograde urohydropropulsion into the bladder and removed by minimally invasive procedures.
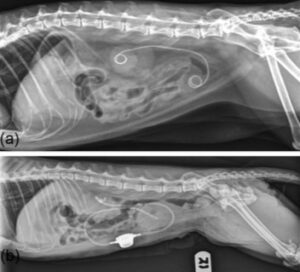
Pic 1: Lateral radiographs of cats treated with a ureteral stent (a) and subcutaneous ureteral bypass device (b)
Treatment of upper urinary tract (kidney and ureter) uroliths
- Nephrolithiasis: Only problematic nephroliths (causing outflow obstruction, renal parenchymal compression, recurrent infections, or pain) require treatment. Nephroliths were not found to significantly affect progression of CKD in cats and the same is likely true for dogs. Suspected struvite nephroliths, which represent about 20-30% of upper urinary tract uroliths should be dissolved. Suspicion is based on alkaline urine, UTI caused by urease-producing bacteria and moderate radioopacity. If ureters are obstructed, they should be stented to allow medicated urine to reach the calculus and for evacuation of bacteria and inflammatory debris. Over 90% of nephroliths and ureteroliths in cats are calcium oxalates, therefore, dissolution is not recommended. Problematic nephroliths should be removed by endoscopic nephrolithotomy or extracorporeal shockwave lithotripsy (applicable only in dogs with nephroliths < 1.5cm). Larger nephroliths require ureteral stenting.
- Hydronephrosis and hydroureter proximal to an obstructive lesion visualized by ultrasound are sufficient to diagnose ureteral obstruction. Partial and complete ureteral obstructions should be managed as an emergency as they can result in a rapid and lasting decrease in renal function. Decompression by subcutaneous ureteral bypass, ureteral stent, or traditional surgery is necessary when medical management fails or is contraindicated in severely ill patients (Pic 1). Interventional procedures have lower morbidity and mortality rates than traditional surgical options, therefore, referral is advised. Renal function can be maximized by relieving obstruction even after a long time (>8 weeks); therefore, the duration of obstruction should not be a deciding factor.
- Medical treatment for obstructive ureteroliths can be considered for 24-72 hours including fluid diuresis, mannitol, alpha adrenergic antagonists and tricyclic antidepressants. Antibiotics should be administered in dogs because of the high incidence of concurrent UTI and pyelonephritis. Medical management is effective in only 8-13% of feline cases.
- Subcutaneous ureteral bypass or ureteral stenting in cats and ureteral stenting in dogs are considered the treatment of choice for ureteral obstruction.
Urolith prevention
Additional therapeutic strategies are needed to prevent urolith recurrence or stent encrustation, based on quantitative urolith analysis.
Dogs, and especially cats with sterile struvites, should be fed acidifying therapeutic diets with low magnesium and phosphorus (goal: pH<6.5). The most important step in prevention of infection-induced struvites is elimination of UTI based on urine culture, as urinalysis is an insensitive marker for UTI. In patients with recurrent UTI, structural and functional risk factors should be investigated.
To minimize calcium oxalate urolith recurrence, high-moisture (>75% water) diets are recommended to achieve USG < 1.020 in dogs and < 1.030 in cats. High sodium diets are not a substitute for high-moisture diets, as their effect is short-lived. Acidifying diets with excessive protein content should be avoided. Potassium citrate can be added in patients with persistently acidic urine. Thiazide diuretics can be considered in patients with a high recurrence rate of these calculi.
Prevention of urate uroliths consists of decreasing urine concentration, limiting purine intake and alkalinizing urine pH. In dogs with genetic mutation in the urate transporter, allopurinol therapy can decrease the high rate of urate recurrence.
Prevention strategies for cystine uroliths consist of decreasing USG, limiting animal protein and sodium intake, increasing urine pH > 7.5, and neutering.
For more information on this topic or Angell-West’s Internal Medicine Service, please contact Dr. Ackley at dvrabelova@angell.org or 781-902-8400
References
- Lulich JP, Berent AC, Adams LG, et al. ACVIM Small Animal Consensus Recommendations on the Treatment and Prevention of Uroliths in Dogs and Cats. J Vet Intern Med 2016;30:1564-1574
- Horowitz C, Berent A, Weisse Ch, et al. Predictors of outcome for cats with ureteral obstructions after interventional management using ureteral stents or a subcutaneous ureteral bypass device. J Feline Med Surg 2013;15(12):1052-1062