-
Adopt
-
Veterinary Care
Services
Client Information
- What to Expect – Angell Boston
- Client Rights and Responsibilities
- Payments / Financial Assistance
- Pharmacy
- Client Policies
- Our Doctors
- Grief Support / Counseling
- Directions and Parking
- Helpful “How-to” Pet Care
Online Payments
Emergency: Boston
Emergency: Waltham
Poison Control Hotline
-
Programs & Resources
- Careers
-
Donate Now
 Maureen C. Carroll DVM, DACVIM (Internal Medicine)
Maureen C. Carroll DVM, DACVIM (Internal Medicine)
www.angell.org/internalmedicine
internalmedicine@angell.org
617-546-5186
For decades scientists have been focusing research endeavors on treating liver fibrosis and cirrhosis in people, and subsequently our knowledge about the treatment options for dogs is based on what we have learned in human medicine. The many inflammatory liver diseases we encounter in canine medicine, when left unchecked can lead to fibrosis and eventual cirrhosis, followed by liver failure. For many dogs hepatic changes associated with these histopathological findings are discovered at the time of liver biopsy, when most feel treatment options are limited. Our goal therefore is to make every therapeutic attempt to prevent further deposition of fibrous tissue, or even reverse the changes that have already occurred. Interestingly the role of the renin- angiotensin aldosterone (RAS) system in the liver and its role in extracellular matrix production in the liver (and other organs) is one that has received attention in the past few years. The role of angiotensin receptor blockers in the management of liver fibrosis will be reviewed, along with other more common medications that can affect fibrous tissue deposition in the liver. Although in the end our efforts in controlling fibrosis in the liver will involve targeting inflammation, copper, oxidative stress; and omitting certain drugs that would cause liver damage, there is data that does support targeting pathways like the RAS in managing these patients.
First, I would like to distinguish between liver FIBROSIS and liver CIRRHOSIS. Liver fibrosis is caused by inflammation in the liver, resulting in activation of stellate cells and subsequent increase in production of extra cellular matrix (ECM). ECM is a collection of molecules secreted by cells that provide structural and biochemical support to the surrounding cells. Collagens are the major proteins in the ECM. Therefore an increase in ECM results in accumulation in fibrous tissue and is a ‘necessary’ pathway in most inflammatory conditions of the liver. When fibrosis advances to the point where portal areas become ‘bridged,’ (you may see ‘bridging fibrosis’ written in many of your liver histopathology reports) the result is the formation of what we call ‘pseudo- lobules’. This finding represents fibrosis in its final stage, cirrhosis.
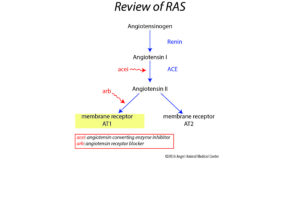
The RAS pathway is reviewed in the diagram here. The receptor targets for Angiotensin II are the angiotensin membrane receptors AT1 and AT2, which have opposing actions upon stimulation. AT1 is involved in vasoconstriction and cellular proliferation, and AT2 is involved in the opposite (vasodilation and anti-proliferation).
Angiotensin II is involved in key events in the inflammatory pathways of the liver. The effect of Angiotensin II on AT1 receptors results in TGF- b1 secretion, which stimulates cell growth, proliferation and differentiation.
As a result, membrane receptor AT1 is the major receptor in the RAS system involved in synthesis and deposition of fibrous tissue. Activated hepatic stellate cells in vivo and in culture express renin and angiotensin-converting enzyme (ACE), and secrete Ang II, which in turn stimulates the AT1 receptors.1 Plasma renin activity and aldosterone levels are increased in cirrhotic liver patients.2 The multitude of data available to us today supports the role the RAS plays in the progression of liver fibrosis.
AT1 receptor blockers (arb/see diagram), have been shown to lessen liver fibrosis in animal models based on blockade of ‘pro fibrogenic’ activity that results when this receptor is stimulated. Some studies in animals even showed reduction in fibrosis using arb’s where the condition was considered advanced.4 In comparison to ACE inhibitors, angiotensin receptor blockers were superior for this function.3
The majority of studies examining the use of arb’s in the treatment of liver fibrosis have involved the use of Losartan, which has been evaluated in hepatitis C, non- alcoholic fatty liver disease, and in various small pilot studies. The results are a decrease in collagen IV deposition as well as TGF-b1 expression, resulting in a lower fibrosis- progression index. Because most studies have been small, larger trials need to be conducted, however initial results in certain patient populations are promising.4
In our hepatitis patients whom we diagnose with fibrosis, I routinely prescribe Losartan (or Telmisartan, see below) at a dose of 0.5 mg/ kg once to twice daily. Ideally we would perform a follow- up liver biopsy 4-6 months after starting this drug and others, but in most instances the owners find a second biopsy cost-prohibitive, or too much to put their animal through procedurally.
Recent pharmacology studies have shown that the metabolism of Losartan is quite different in humans compared to dogs. Dogs metabolize only a small percentage of losartan into the active metabolite, which is responsible for most of the therapeutic effects. The pharmacology profile of Telmisartan in dogs is superior, and is becoming the arb of choice in this species. Dosage is the same as Losartan. Monitoring includes following trends in blood pressure, electrolytes, and renal function.
Other drugs useful in veterinary medicine to target fibrosis include corticosteroids (will not be reviewed here), zinc compounds, as well as chelating agents like penicillamine. All of these drugs have antifibrotic effects, and can in theory slow the progression of fibrosis in inflammatory liver disease.
Zinc acetate is a drug I use often in patients with either copper hepatopathy (following the use of either penicillamine or Trientine) or in animals with fibrosis/bridging fibrosis secondary to inflammatory liver disease. The dose for hepatic fibrosis is 15 mg/kg/day of ELEMENTAL zinc, administered on an empty stomach. Average dose of zinc acetate for a medium dog is 200 mg/day. Trough Zinc levels should be monitored every 2-4 months to guard against toxicity. Target plasma level is between 200-400 mcg/dl.
Penicillamine and Trientine are chelating agents that are used most commonly for Copper storage disease in dogs. Anti- fibrotic activity is based on this drug’s ability to inhibit collagen cross-linking resulting in increased collagen degradation. The standard dose is 10- 15 mg/kg BID for chelation. Doses necessary to treat chronic hepatitis and fibrosis are likely higher, and side effects (nausea and vomiting) may preclude its use. Trientine has a lower side effect profile compared to Penicillamine, dose is 5-7 mg/kg BID.
Colchicine is a drug that has been used in treating people with chronic hepatitis and liver fibrosis, but there is still a lack of convincing evidence in both humans and animals to warrant its use for liver disease.
In summary, targeting inflammation, cytokine upregulation, oxidative damage and potentially hepatotoxic drugs is our best defense in preventing and managing the progression of liver fibrosis in our canine patients. Addressing the RAS system is a newer modality in veterinary medicine in managing fibrotic diseases of the liver, as the research performed in humans and lab animals is promising. More research is clearly necessary in dogs before we can firmly recommend the use of these drugs. However because of the low toxicity profile in general of angiotensin receptor blockers, empiric use at this time is appropriate in my opinion.
For more information, please contact Angell’s Internal Medicine service at 617-541-5186 or internalmedicine@angell.org.
References
- Bataller R, Sancho- Bru P, Gines P et al. Activated human hepatic stellate cells express the renin- angiotensin system and synthesize angiotensin II. Gastroenterology 2003; 125; 117-25
- Bosch J, Arroyo V, Betriu A et al. Hepatic hemodynamics and the renin- angiotensin- aldosterone system in cirrhosis. Gastroenterology 1980; 78 92-9
- Kim MY, Baik SK, Park DH et al. Angiotensin receptor blockers are superior to angiotensin- converting enzyme inhibitors in the suppression of hepatic fibrosis in a bile duct- ligated rat mode. J Gastro 2008; 43: 889-96
- K. Munshi, J. Uddin, S. Glaser ‘The role of the renin- angiotensin system in liver fibrosis’, Experimental Biology and Medicine 2011; 00: 1-9. DOL: 10.1258
- Shinanomachi, S., Namiki, TS, et al.
‘Effect of Telmisartan or Losartan for Treatment of Nonalcoholic Fatty Liver Disease: Fatty Liver Protection Trial by Telmisartan or Losartan Study (FANTASY)’; International Journal of Endocrinology,Volume 2013 (2013) - S. Yokohama, M. Yoneda, M. Haneda et al., “Therapeutic efficacy of an angiotensin II receptor antagonist in patients with nonalcoholic steatohepatitis,” Hepatology, vol. 40, no. 5, pp. 1222–1225, 2004.
- S. Yokohama, Y. Tokusashi, K. Nakamura et al., “Inhibitory effect of angiotensin II receptor antagonist on hepatic stellate cell activation in non-alcoholic steatohepatitis,” World Journal of Gastroenterology, vol. 12, no. 2, pp. 322–326, 2006.
- Plumb, DC. Plumb’s Veterinary Drug Handbook, Eighth Edition John Wiley & Sons, Inc, 2015